9 Advantages of Laboratory Information Management System
Table Of Content
Published Date :
26 Feb 2026
Key Takeaways
- A structured laboratory information management system strengthens compliance, reduces audit risk, and improves operational control.
- Automation lowers manual errors, accelerates turnaround time, and protects revenue margins.
- Integration with EHR and billing systems eliminates data silos and improves workflow efficiency.
- Real-time dashboards provide clear visibility into performance and financial metrics.
- Scalable digital infrastructure supports multi-site growth and long-term strategic expansion.
Healthcare laboratories face multiple challenges related to their operational requirements and their need to comply with regulations. The laboratory system experiences higher test demands while compliance requirements become more stringent. Diagnostic labs require a laboratory information management system to function effectively in this situation.
The diagnostic processes of hospital networks and specialty labs become inefficient because of their disjointed operations. These operations lead to extended reporting times and unaccounted expenditures which negatively affect their profit margins.
The leadership team of an organization must understand the advantages of laboratory information management system adoption because this knowledge enables them to make long-term strategic decisions instead of making short-term operational choices.
What Is Laboratory Information Management System
The first step to assess the impact of laboratory information management system technology requires us to establish its function as a business tool.
A laboratory information management system is a centralized digital platform that manages samples, workflows, data, compliance records, and reporting within a laboratory environment. Labs follow structured workflows which enable them to track every specimen from its accessioning stage until its final reporting stage.
This process requires US diagnostic laboratories to implement barcode tracking systems which automatically validate test results and maintain secure data storage that meets HIPAA regulations while connecting to electronic health record systems.
The system creates operational efficiency by reducing friction which enables executives to monitor real-time metrics, and regulatory compliance status. Laboratories use improved visibility to grow their operations and enhance their market competitiveness.
Looking to Improve Compliance and Control?
Strengthen audit readiness, secure patient data, and streamline workflows with a scalable laboratory information management platform.
Business Benefits of Laboratory Information Management Systems
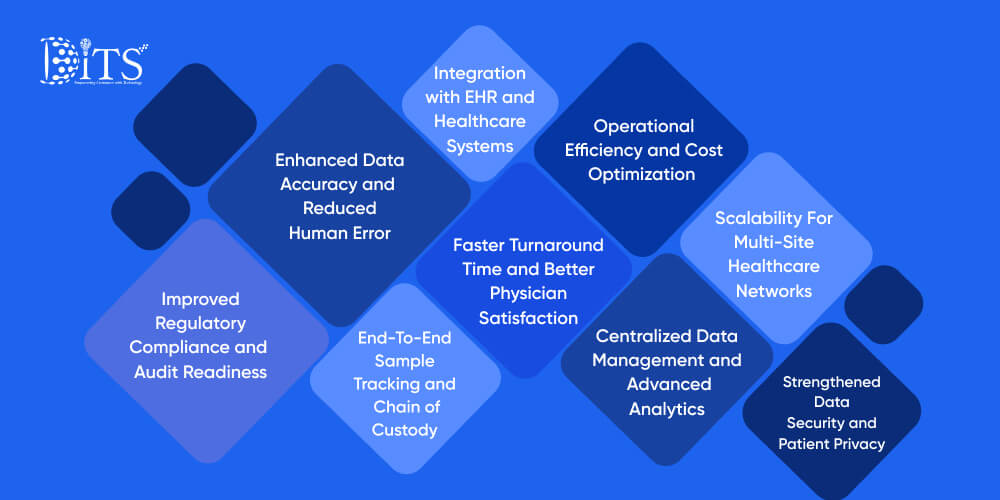
Now that we have an idea of laboratory information management systems and their need in laboratories and healthcare organizations, let us explore their advantages.
1. Improved Regulatory Compliance and Audit Readiness
In U.S., inspections are common. CLIA audits, CAP reviews, HIPAA scrutiny. Documentation gaps can quickly escalate into financial penalties or operational disruption.
One of the strongest advantages of laboratory information management system adoption is structured compliance control. Every sample movement is logged. Every modification is timestamped. Every user action is recorded.
Instead of assembling paperwork weeks before an inspection, labs generate reports in minutes. Here’s the practical impact:
- Automated documentation aligned with CLIA and CAP standards
- Role-based access control to protect PHI
- Secure data retention policies
- Real-time validation rules to prevent non-compliant entries
Compliance stops being reactive and becomes embedded into daily operations. That shift alone reduces risk exposure significantly.
2. Enhanced Data Accuracy and Reduced Human Error
Manual data entry remains one of the most expensive blind spots in laboratory operations. A single transcription error can trigger retesting, delayed billing, or physician dissatisfaction.
With barcode scanning, automated validation rules, and direct instrument integration, the margin of error drops sharply. Labs that previously managed 2 to 3 percent correction rates often reduce that below 0.5 percent within months of implementation.
Consider a regional diagnostic center processing 3,000 samples per day. Even a 1 percent error rate translates to 30 problem cases daily. Multiply that by rework cost, staff time, and client trust. The financial impact adds up quickly.
By systemizing workflows through a laboratory information management system, accuracy becomes predictable rather than dependent on manual oversight.
3. Faster Turnaround Time and Better Physician Satisfaction
Speed matters. Physicians expect results within defined windows, and patients expect quick solutions, but delays affect treatment decisions.
Automated routing, advanced queue management, and real-time status updates reduce bottlenecks. Instead of samples sitting in trays waiting for manual assignment, tasks are digitally distributed based on priority and workload.
Operational improvements often include:
- Automated test assignment
- Priority-based workflow routing
- Instant digital result transmission to EHR systems
- Real-time dashboards for lab managers
Many labs report turnaround improvements between 15 to 30 percent after optimization. That translates directly into stronger provider relationships and higher retention rates.
Faster reporting often improves cash flow because billing cycles begin earlier.
4. End-To-End Sample Tracking and Chain of Custody
Lost samples are rare but costly. Misidentified specimens are worse.
End-to-end tracking ensures every specimen has a digital footprint from collection to disposal. Multi-location networks particularly benefit because samples often travel between facilities.
Core tracking capabilities include:
- Unique barcode identifiers
- Location tracking within facility
- Time-stamped status updates
- Cross-site visibility
For growing diagnostic chains, this visibility strengthens operational control while reducing liability exposure. In competitive markets, that level of reliability builds reputation.
5. Integration with EHR and Healthcare Systems
A modern laboratory information management system integrates with EHR, billing platforms, hospital information systems, and third-party diagnostic tools through HL7 or API-based connections.
For executives, the benefit is operational cohesion. Revenue cycles align. Clinical teams receive real-time updates. Reporting accuracy improves across departments.
Organizations investing in structured healthcare software development services often prioritize this integration layer to eliminate silos and future-proof expansion plans. Because as networks grow, disconnected systems become expensive very quickly.
6. Operational Efficiency and Cost Optimization
Automation reduces administrative burden across accessioning, reporting, billing coordination, and quality checks. Labs that once required manual reconciliation at day’s end often move toward automated validation workflows.
Common efficiency gains include:
- Reduced duplicate testing
- Lower rework rates
- Optimized staff allocation
- Shorter billing cycles
One mid-sized pathology lab reported annual savings exceeding $250,000 after workflow automation reduced overtime and manual corrections. That type of return shifts technology investment from cost center to profit driver.
Organizations working with structured LIMS development services can design workflows that reflect their specific operational model instead of using rigid templates.
7. Centralized Data Management and Advanced Analytics
Laboratories generate enormous volumes of data. Yet without structured analysis, most of it remains unused.
Centralized dashboards allow leadership to monitor key metrics such as average turnaround time, and rejection rates. Instead of relying on static monthly reports, executives gain live operational visibility.
Here is a simplified comparison:
| Metric Visibility | Manual Systems | Digital Platform |
| Real-Time TAT Tracking | Limited | Instant Dashboard |
| Error Trend Analysis | Manual Review | Automated Alerts |
| Multi-Site Reporting | Fragmented | Centralized View |
| Revenue Insight | Delayed | Integrated Reporting |
This level of transparency enables faster decisions. Expansion planning becomes data-driven. Resource allocation becomes strategic.
At DITS, we integrate AI within our development lifecycle to enhance software performance, streamline quality assurance, maintain code integrity, and enable intelligent customization. As an AI software development company, we embed intelligent capabilities into every system to ensure long-term scalability without compromising compliance.
8. Scalability For Multi-Site Healthcare Networks
Growth often exposes weaknesses in legacy systems. What works for one facility rarely scales smoothly to five or ten.
A flexible architecture supports multi-location standardization while allowing site-specific configuration. Test catalogs can be shared. Quality controls remain consistent. Leadership gains centralized oversight without micromanaging each site.
Healthcare groups expanding through acquisitions particularly benefit from unified platforms. Integration becomes structured instead of chaotic.
For organizations launching new service lines or pilot labs, structured MVP development approaches allow phased implementation before full-scale rollout. That reduces risk while validating operational assumptions.
9. Strengthened Data Security and Patient Privacy
Healthcare data is among the most sensitive categories of information. Breaches damage reputation and invite regulatory consequences.
Modern systems implement encrypted storage, secure transmission protocols, and strict access controls. Activity logs provide accountability. Role-based permissions restrict exposure.
Instead of relying on shared credentials or unsecured files, security becomes embedded within architecture. This reduces internal risk and strengthens external compliance posture.
Planning Multi-Site Laboratory Expansion?
Deploy scalable digital infrastructure that standardizes workflows while maintaining flexibility across facilities and service lines.
How LIMS Supports Strategic Growth
When evaluating the best laboratory information management system, leadership should think beyond operational convenience. The real value lies in long-term positioning.
Structured systems support:
- Value-based care reporting
- Expansion into specialty diagnostics
- Data-driven pricing models
- Improved provider retention
Labs become strategic assets rather than transactional service units.
At DITS, our approach to LIMS Development Services aligns technology architecture with compliance requirements and growth objectives. We integrate AI intelligently into development, testing, and customization processes to deliver systems that adapt as healthcare markets evolve.
Why Choose DITS For Laboratory Information Management Software
Selecting a technology partner for laboratory information management software is not just a procurement decision. It is a long-term operational commitment. The right partner understands healthcare regulations, integration complexity, and the financial pressure laboratories face every quarter.
At DITS, we approach every laboratory information management system implementation as a business transformation initiative rather than a standalone software project. Our teams work closely with lab directors, CIOs, and compliance officers to map workflows before writing a single line of code.
What differentiates us is structured engineering discipline combined with intelligent automation. We use AI internally across software development, quality assurance, code review, and customization processes to maintain performance stability and long-term scalability. This ensures cleaner architecture, fewer defects, and faster deployment cycles without compromising regulatory alignment.
Our strengths include:
- Custom-built platforms aligned with CLIA, CAP, and HIPAA requirements
- Seamless EHR and billing integration through proven interoperability frameworks
- Scalable multi-site architecture for growing healthcare networks
- Data analytics capabilities that convert lab activity into executive insight
- Phased rollout strategies to minimize operational disruption
For healthcare organizations evaluating the best laboratory information management system, the decision should balance technology depth with domain expertise. DITS combines both, ensuring your lab operations are efficient today and strategically prepared for tomorrow.
Conclusion
Healthcare laboratories in the United States operate under increasing financial, operational, and regulatory pressure. Technology investments must deliver measurable returns.
The advantages of laboratory information management system adoption extend far beyond workflow automation. They influence compliance readiness, cost control, physician satisfaction, scalability, and strategic growth.
For executive teams, the question is no longer whether digital systems are necessary. It is how quickly they can deploy a structured, secure, and scalable solution that supports long-term competitiveness.
Frequently Asked Questions
What is laboratory information management system and why is it important for healthcare businesses?
A laboratory information management system is a digital platform that manages lab workflows, sample tracking, compliance documentation, and reporting. For healthcare businesses in the U.S., it improves operational control, reduces manual errors, and ensures alignment with regulatory standards such as CLIA and HIPAA. It also supports faster turnaround time and better financial visibility.
How long does it take to implement laboratory information management software?
Implementation timelines depend on lab size, workflow complexity, and integration needs. A mid-sized diagnostic lab may require three to six months for full deployment, including data migration and user training. Larger multi-site healthcare networks may require phased rollouts to minimize disruption.
Can DITS customize LIMS software for multi-site healthcare networks?
Yes. DITS LIMS software development services are designed to support scalable, multi-location laboratory environments. We build custom workflows, centralized dashboards, and secure integration layers that allow leadership teams to manage multiple facilities under a unified system while maintaining site-specific flexibility.
How does LIMS improve revenue and cost control?
A structured system reduces rework, prevents duplicate testing, shortens billing cycles, and minimizes compliance-related penalties. Real-time reporting also helps management identify underperforming test categories or operational bottlenecks, enabling faster corrective decisions that protect margins.
Why should healthcare organizations consider DITS LIMS software development services?
Off-the-shelf tools often limit customization and integration flexibility. DITS LIMS software development services focus on building platforms aligned with your exact workflow, regulatory needs, and growth strategy. Our approach integrates intelligent automation, robust quality assurance, and long-term scalability so your investment continues to deliver measurable business value.

Nidhi Thakur
With more than 19 years of experience - I represent a team of professionals that specializes in the healthcare and business and workflow automation domains. The team consists of experienced full-stack developers supported by senior system analysts who have developed multiple bespoke applications for Healthcare, Business Automation, Retail, IOT, Ed-tech domains for startups and Enterprise Level clients.
Recent Posts

Discover the key medical billing software requirements your practice needs. From HIPAA compliance to claims management, understand what to look for.

Discover how the right medical billing software features can simplify your workflow, get you paid faster, and boost your bottom line. Learn about must-have functionalities today.

Build feature rich mental health applications with Ditstek Innovations. We are a leading mental health app development company developing apps for clients worldwide.