Guide to Build Clinical Trial Financial Management Software
Table Of Content
Published Date :
23 Feb 2026
Key Takeaways
- Financial visibility directly impacts trial profitability and compliance.
- A connected system reduces payment delays and reconciliation errors.
- Built-in regulatory controls protect against audit risk.
- Real-time dashboards support faster executive decision-making.
- Strong integration and scalable architecture enable long-term growth.
- Purpose-built platforms create operational efficiency across multi-site trials.
Clinical trials are expensive, complex, and highly regulated. A single late-stage study can involve 40 to 60 sites, multiple vendors, investigator payments, startup fees, and changing protocol requirements. When financial tracking is not structured, small errors multiply quickly. That is why clinical trial financial management software is becoming essential for sponsors, CROs, and research organizations that want predictable margins.
Business leaders today are not only focused on scientific success. They are responsible for financial discipline, audit readiness, and accurate reporting. Spreadsheets cannot handle constant amendments. Traditional accounting systems do not understand research billing rules. And financial surprises during an audit are the last thing any executive wants. A dedicated system brings clarity before problems escalate.
U.S. Clinical Trial Financial Environment
Financial relationships in clinical research are layered. Sponsors fund trials. CROs manage operations. Sites conduct visits and expect timely payments based on agreed contracts. Every visit, procedure, and milestone has a financial impact.
Now add Medicare Coverage Analysis, Sunshine Act reporting, IRB budget approvals, and HIPAA compliance. Each requirement affects how payments are tracked and documented. If finance teams use separate tools for budgeting, contracts, visit tracking, and accounting, reconciliation becomes slow and error-prone.
Many executives discover financial gaps only at quarter-end reviews. By then, correcting errors takes time and money. A connected system prevents these blind spots and keeps leadership informed in real time.
Core Objectives of Clinical Trial Financial Management Software
The goal of a well-designed system is simple: connect budgets, contracts, visits, and payments into one controlled platform. When a subject visit is completed and verified, payment logic should follow automatically. When enrollment changes, financial forecasts should adjust instantly.
Leadership should be able to see:
- Study budget versus actual spend
- Outstanding site payments
- Revenue recognition timelines
- Cash flow projections
This is not just automation. It is control. With strong visibility, executives can make decisions early instead of reacting late.
Are Your Clinical Trial Finances Truly Under Control?
Gain structured visibility across budgets, payments, and compliance with a purpose-built financial platform designed for scalable clinical growth.
Must-Have Features for U.S.-Focused Solutions
A U.S.-focused healthcare software development company helps with clinical trial financial system that must support flexible budgeting, contract tracking, automated payments, and strong compliance controls. It should integrate with core platforms, provide real-time reporting, and follow a secure, scalable architecture designed to meet strict regulatory requirements from the start.
Budget Planning and Scenario Modeling
Clinical trials rarely go exactly as planned. Enrollment may slow. Sites may request rate adjustments. Protocol amendments may change visit schedules. The system must allow budget updates without losing previous versions.
Finance leaders should be able to compare forecasted costs with real spending at any time. Scenario planning tools help answer questions like, “What happens if enrollment drops by 10 percent?” Clear answers reduce uncertainty.
Contract And Grant Management
Contracts define how and when sites get paid. These agreements change over time, especially during long studies. The software should track every amendment clearly.
Automated payment triggers tied to verified visits reduce manual review. Transparent contract history prevents disputes. Reliable payments also strengthen relationships with research sites.
Payment Processing and Reconciliation
Payment delays often happen because finance teams manually verify visit data before releasing funds. By integrating with CTMS and EDC systems, payments can be triggered automatically once visits are confirmed.
Built-in checks should prevent duplicate payments and billing errors. Faster and accurate payouts improve site satisfaction and reduce administrative workload.
Compliance And Audit Readiness
Regulatory pressure in United States is constant. Systems must support audit trails, CMS billing validation, and 21 CFR Part 11 requirements.
Compliance should not depend on manual documentation. When financial systems automatically log every action, audit preparation becomes structured and predictable instead of stressful.
Integration Capabilities
Financial systems cannot work in isolation. They must connect with CTMS, EDC, ERP, and EHR platforms. Smooth integration ensures that operational data and financial data remain aligned.
Strong Enterprise Software Development practices are critical here. Without disciplined integration, even the best-designed system will create data silos instead of clarity.
Advanced Reporting and Dashboards
Executives do not want raw spreadsheets. They want insights. Clear dashboards should show profitability by study, payment cycle performance, and budget utilization trends.
When leadership can see numbers instantly, decision-making speeds up. Portfolio adjustments become proactive rather than reactive.
Technical Architecture Considerations
Scalability matters. Many organizations choose cloud-based deployment because it supports multi-site trials and remote collaboration. Secure Cloud Computing Services provide flexibility while maintaining data protection standards.
However, security cannot be secondary. Systems must include encrypted storage, controlled access permissions, and strong validation testing. Structured QA Software Testing Services ensure performance stability and regulatory readiness before launch.
Choosing the right architecture early prevents expensive redesign later.
Regulatory And Compliance Framework In USA
Financial platforms must align with HIPAA, FDA guidelines, 21 CFR Part 11, and sometimes SOC 2 controls. Some states also have stricter privacy requirements.
Compliance should be built into the system from the beginning. Retrofitting regulatory controls later increases cost and risk. A compliance-first design approach protects both revenue and reputation.
Step-By-Step Roadmap to Build Clinical Trial Financial Management Software
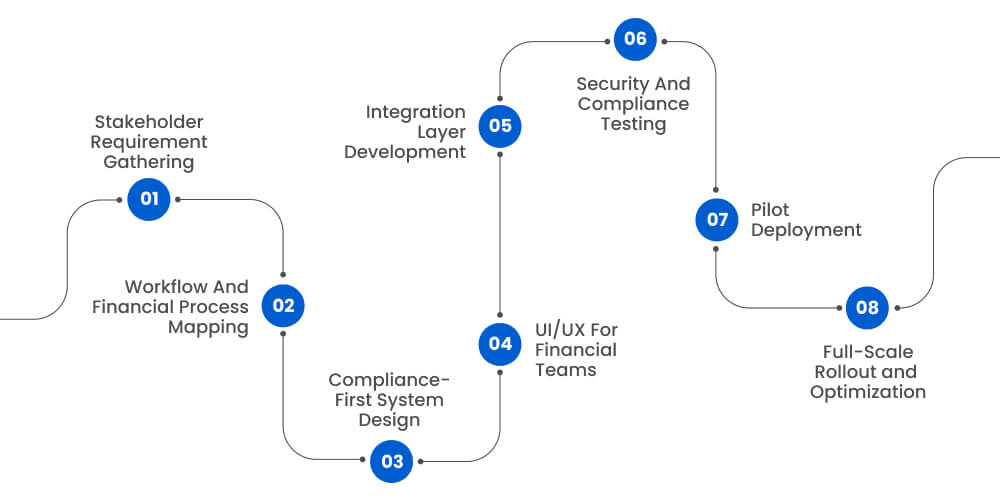
A structured approach to building clinical trial financial management software ensures alignment between stakeholders, compliance requirements, and operational workflows. By following step-by-step implementation, organizations can reduce financial risk, improve reporting accuracy, and achieve scalable, audit-ready financial control across clinical trials.
Step 1: Stakeholder Requirement Gathering
Development begins with alignment among finance leaders, clinical operations teams, compliance officers, and IT stakeholders. Each group identifies current bottlenecks, reporting gaps, and risk exposure points. Clear requirement definition prevents scope drift and protects investment value.
Step 2: Workflow And Financial Process Mapping
Organizations must document real-world processes across budgeting, contracting, visit validation, and payment authorization. Mapping these workflows reveals inefficiencies and clarifies automation opportunities. Process visibility ensures software logic mirrors operational reality.
Step 3: Compliance-First System Design
Regulatory obligations should guide architecture decisions. Access controls, audit logging mechanisms, and validation frameworks must be embedded during system design rather than layered later.
Step 4: UI/UX For Financial Teams
User interfaces should prioritize clarity over visual complexity. Financial analysts require intuitive dashboards, drill-down reporting, and streamlined approval workflows. Adoption improves when tools reflect user behavior patterns.
Step 5: Integration Layer Development
Secure API connections link CTMS, EDC, ERP, and accounting platforms. Synchronization eliminates data silos and supports real-time financial visibility.
Step 6: Security And Compliance Testing
Comprehensive testing validates system stability, security posture, and regulatory readiness. Structured validation documentation supports audit transparency.
Step 7: Pilot Deployment
Controlled rollout within select studies enables real-world feedback and refinement. Early issue detection prevents broader disruption.
Step 8: Full-Scale Rollout and Optimization
After stabilization, deployment expands across study portfolios. Ongoing optimization enhances forecasting accuracy and reporting sophistication.
Want Real-Time Visibility Into Trial Profitability?
Transform fragmented financial data into executive dashboards that support faster decisions and predictable study margins.
Common Challenges Businesses Face
Many organizations struggle with delayed site payments, budget mismatches, and manual reconciliation errors. Limited visibility makes it difficult to track margin performance across multiple trials. In worst cases, billing compliance gaps lead to penalties.
These issues are often symptoms of fragmented it financial management software that was not designed specifically for clinical research. A structured system reduces these risks significantly.
Cost Factors and ROI Expectations
Development costs depend on customization depth, integration complexity, compliance validation requirements, and infrastructure scale. Initial investment includes architecture design, testing protocols, and security certifications. However, operational savings typically emerge within the first year of deployment. With Ditstek Innovations, the cost to build clinical trial financial management software starts with $25,000 or $22-25/hr with the rapid development procedure and transparent communication.
The following table outlines common cost drivers and measurable impact areas:
| Cost Driver | Operational Impact | Long-Term Value |
| Custom Development Scope | Tailored workflow automation | Reduced manual labor |
| Integration Complexity | Real-time data synchronization | Faster decision cycles |
| Compliance Validation | Audit readiness assurance | Lower regulatory risk |
| Infrastructure Investment | Scalable performance | Portfolio expansion capability |
For mid-sized CROs managing multiple concurrent trials, even modest reductions in reconciliation time and payment disputes generate measurable financial return. Structured cloud financial management software environments also enable scalable expansion without structural redesign.
Why Choose DITS For Clinical Trial Financial Management Software
DITS delivers structured product engineering services designed for long-term scalability and compliance alignment. Our enterprise software development expertise ensures secure integration across research and accounting systems.
We also apply Artificial Intelligence in Healthcare thoughtfully within financial platforms. AI supports anomaly detection, forecasting, and automation. Internally, we use AI for software development acceleration, code quality monitoring, testing efficiency, and solution customization. This approach strengthens reliability without adding unnecessary complexity.
Through disciplined QA software testing services and secure cloud computing services, we build resilient systems that support growth and regulatory confidence.
Struggling With Clinical Billing Compliance Risks?
Build a compliance-first financial architecture that strengthens audit readiness while improving financial transparency across multi-site trials.
Conclusion
Clinical research finance is no longer a back-office task. It directly affects profitability, compliance standing, and strategic growth. Organizations that rely on disconnected tools often face surprises at the worst possible time.
By investing in structured clinical trial financial management software, business leaders gain real-time visibility, predictable margins, and stronger operational control. Financial clarity supports smarter decisions. And smarter decisions drive long-term success.
Frequently Asked Questions
What Is Clinical Trial Financial Management Software and Why Do Organizations Need It?
Clinical trial financial management software is a specialized system designed to manage study budgets, contracts, investigator payments, billing compliance, and revenue tracking in one connected platform. Organizations need it to avoid payment delays, reduce reconciliation errors, and maintain audit readiness. Without a structured system, financial visibility becomes fragmented, especially in multi-site studies.
How Does Clinical Trial Financial Management Software Improve Cash Flow Control?
It connects subject visit data, contract milestones, and payment schedules in real time. When visits are verified, payments can be triggered automatically based on predefined rules. This reduces manual processing time and gives executives clearer insight into budget utilization, outstanding payments, and projected revenue timelines.
How Long Does It Take to Develop a Custom Financial Platform for Clinical Trials?
Development timelines typically range from six to twelve months depending on integration complexity, compliance requirements, and customization scope. Projects that require integration with CTMS, EDC, and ERP systems may take longer. A phased rollout approach often accelerates adoption while minimizing operational disruption.
How Does DITS Clinical Financial Software Development Services Support Regulatory Compliance?
DITS clinical financial software development services are designed with compliance-first architecture. Our approach ensures systems align with HIPAA, 21 CFR Part 11, and CMS billing requirements from the initial design stage. We embed audit trails, validation documentation, and secure access controls within the platform to reduce regulatory exposure and simplify audit preparation.
Can AI Be Integrated into Clinical Trial Financial Systems?
Yes. AI can enhance forecasting accuracy, detect anomalies in payment data, and automate reconciliation processes. Through DITS clinical financial software development services, AI capabilities are thoughtfully integrated into financial platforms while maintaining strict compliance standards. We also use AI internally to strengthen development quality, testing efficiency, and solution customization.
What Is the Return On Investment for a Dedicated Financial Platform?
Return on investment becomes visible through faster payment cycles, fewer billing errors, improved compliance readiness, and reduced administrative workload. For organizations managing multiple concurrent trials, even small efficiency improvements can generate significant annual savings while strengthening executive-level financial oversight.

Nidhi Thakur
With more than 19 years of experience - I represent a team of professionals that specializes in the healthcare and business and workflow automation domains. The team consists of experienced full-stack developers supported by senior system analysts who have developed multiple bespoke applications for Healthcare, Business Automation, Retail, IOT, Ed-tech domains for startups and Enterprise Level clients.
Recent Posts

Learn the benefits of AI in manufacturing industry for predictive maintenance, automation, quality control, supply chain optimization, and scalable data-driven manufacturing operations.

A typical mobile app development team includes key roles such as Project Manager, UI/UX Designer, Mobile App Developer, and QA Engineer.

Learn the benefits of using AI in education for personalized learning, automated assessments, student engagement, academic analytics, and scalable digital learning environments.